BIREME collaborates with the development of the Regional Database of Health Technology Assessment Reports (BRISA), coordinated by the Medicines and Health Technologies Unit of the Department of Health Services and Systems (MT/HSS) of the Pan American Organization of Health/ World Health Organization (PAHO/WHO).
The reports included in the BRISA database are produced by national institutions, members of the Health Technology Assessment Network of the Americas (Red de Evaluación de Tecnologías en Salud de las Américas, RedETSA) in Argentina, Brazil, Canada, Chile, Colombia, Costa Rica, Cuba, El Salvador, Ecuador, Mexico, Peru, United States, and Uruguay. This initiative aims at developing new information services for BRISA, as well as the integration with the RedETSA website
In 2017, the portal http://sites.bvsalud.org/redetsa/brisa was launched during the IX RedETSA Meeting, held in Panama City, which had then 600 reports published in the BRISA database. To date, BRISA offers access to around 2,500 reports, of which 280 are related to the SARS-CoV-2 virus and COVID-19.
 With the growing number of reports indexed in the database, associated with the increased visibility and use of BRISA, it became necessary to develop new functionalities that further promote the access and use of available information for health decision-making.
With the growing number of reports indexed in the database, associated with the increased visibility and use of BRISA, it became necessary to develop new functionalities that further promote the access and use of available information for health decision-making.
At the same time, the necessity to update and renew the RedETSA portal was identified, preserving the Network’s historical information, expanding the integration between the RedETSA and BRISA portals, promoting the development of new functionalities for users, such as user profiles and toolkit.
Within this context, the following resources were published:
- RedETSA Portal with a renewed technological platform, information architecture, and visual identity.
- Subportal with the contents of the BRISA database and topics related to COVID-19, developed to facilitate access to evaluation reports through pre-established search strategies.
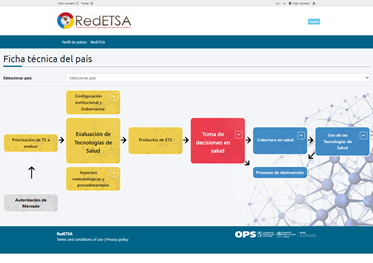
- Subportal of technical files from developed countries to disseminate the state of the art of RedETSA members in terms of health technology assessment.
- HTA toolkit Subportal, designed to expand its use by emerging countries.
 To keep the information always dynamic and updated, BIREME, with feedback from partner institutions, periodically adjusts the content of the portals, according to the needs identified.
To keep the information always dynamic and updated, BIREME, with feedback from partner institutions, periodically adjusts the content of the portals, according to the needs identified.